Measuring the pH of hydrothermal fluids at high temperature and high pressure: a challenge in geology and many industrial applications
Geologists, chemists and geochemists of the GeoResources laboratory (CNRS, University of Lorraine), of the IRAP (CNRS, University of Toulouse) and of the IGEM (Russian Academy of Sciences) have succeeded in measuring, in laboratory, the pH of hydrothermal fluids in water-gas-salts systems up to 280° C and 150 bar pressure. The device, miniaturized, robust and accurate, not only allow to measure the pH in extreme conditions, but also to equip industrial installations implementing hot and pressurized fluids: oil drilling platforms, geothermal power stations, cooling circuits of nuclear power stations.

The pH or hydrogen potential is a measure of the activity of H+ ions in solution. It is a measure of the acidity or basicity of a solution. It is a key parameter, classically measured in chemistry, whether for environmental monitoring, monitoring of an industrial process, or more generally for studying reactions in solution.
In geology, water-rock-gas interactions are at the heart of many natural processes, whether terrestrial or extraterrestrial. Their understanding and modeling generally go through a reproduction phase in the laboratory, which is often a critical step in establishing and validating the models that describe or predict them. In the high-temperature range, between the boiling point and the critical point of water (100-373°C), liquid water is only stable in a confined environment at high pressure. The measurement of its physico-chemical properties requires the cooling of a sample assumed to be representative of the system. Most of the dissolved ions are not affected by this operation. However, this is not the case for pH and sometimes redox potential, which are nevertheless essential variables for studying the chemical reactivity of the medium.
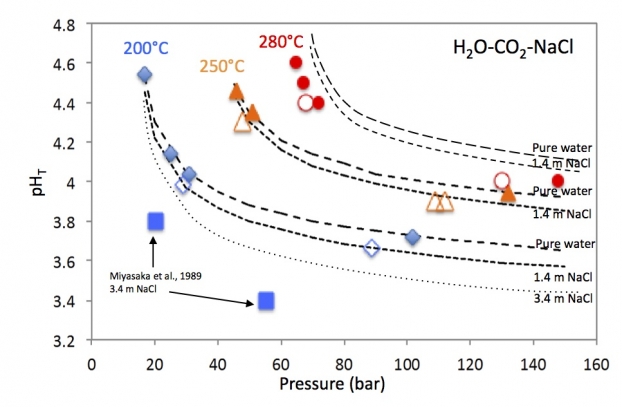
Measuring pH in hydrothermal conditions is a major instrumental challenge. While it is true that these measurements are easily performed at temperatures below 150°C using glass potentiometric probes, the field of more severe hydrothermal conditions remains the preserve of a few rare laboratories in the world. The very heavy devices used by these laboratories prevent routine measurements from being carried out and therefore remain confidential. They are, moreover, unsuitable for fluid-gas mixtures. By optimising a reference probe and a pH probe made of ceramic (yttrium-doped zirconia), these researchers have managed to considerably increase the field of use of a commercial device and to couple it to a high-pressure, high-temperature autoclave allowing both the sampling of fluids and gases and in-situ measurements by Raman spectroscopy. The result is an innovative device allowing continuous pH measurements to be carried out up to 280°C and 150 bar, while having access to in-situ speciation of chemical elements by Raman spectroscopy, and to the composition of the fluid, gases and rocks by means of sampling.

By using the H2O-CO2-NaCl system as a reference for testing their instruments under extreme conditions, the researchers were able to demonstrate their reliability and high measurement accuracy. The coupling of potentiometry and in-situ Raman spectroscopy allows for the first time to control all system variables in real time. The novelty of these measurements has also enabled them to develop a versatile thermodynamic model that can be used by a large community of researchers and engineers.
The development of this equipment was part of a Défi Instrumentations aux Limites project funded by the CNRS. It could also go beyond the laboratory setting to integrate industrial facilities where high-temperature and high-pressure aqueous fluids are used, such as the cooling circuits of nuclear power plants or oil drilling platforms, in order to improve safety and continuously monitor the properties of the fluids.
Laboratories involved
- Le laboratoire GeoRessources (CNRS/Université de Lorraine/CREGU) ;
- L’Institut de Recherche en Astrophysique et Planétologie (CNRS/Université de Toulouse) ;
- L’Institut de Géologie des Gisements de Métaux (IGEM/Académie des Sciences de Russie), Moscou, Russie.
Source(s):
- Direct measurement of CO2 solubility and pH in NaCl hydrothermal solutions by combining in-situ potentiometry and Raman spectroscopy up to 280°C and 150 bar.Truche, L., Bazarkina, E., Bessaque, G., Berger, G., Caumon M.-C., Dubessy, J. Geochimica and Cosmochimica Acta (GCA), 30 janvier 2016. http://www.sciencedirect.com.biblioplanets.gate.inist.fr/science/article/pii/S001670371600048X
IRAP Contact
- Gilles Berger, gilles.berger@irap.omp.eu






